Delhi: The Central Drugs Standard Control Organisation (CDSCO) has launched a probe after the World Health Organisation issued an alert against four India-made cold and cough syrups that have been “potentially linked” with acute kidney injuries and 66 deaths among children in the African country, The Gambia.
According to government sources, CDSCO has taken up the matter and ordered a detailed investigation against the cough and cold syrups produced and exported to The Gambia by Maiden Pharmaceuticals Limited, which is based in Haryana’s Sonepat, on the basis of available information shared by WHO.
Notably, the World Health Organization on Wednesday issued an alert against four cough and cold syrups made by India-based Maiden Pharmaceuticals, saying that it might be linked to the deaths of 66 minor children in the Gambia. The international health body reportedly said that the cough and cold syrups have ingredients, such as diethylene glycol and ethylene glycol, which can be toxic for human beings.
The global body said it is conducting further investigation with the company and Indian regulatory authorities.
As per reports, last month the Gambian government reported that it has been investigating the deaths of small children under the age of 5 years as there was a spike in cases of acute kidney injury, which surfaced in July.
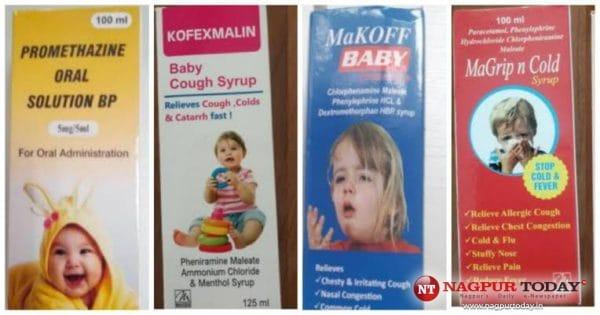
The four syrups that have been identified are Promethazine Oral Solution, Kofexmalin Baby Cough Syrup, Makoff Baby Cough Syrup, and Magrip N Cold Syrup, as per news reports.
“Laboratory analysis of samples (in the syrup) of each of the four products confirms that they contain unacceptable amounts of diethylene glycol and ethylene glycol as contaminants,” the WHO said in a medical product alert.
In a tweet, Tedros Adhanom Ghebreyesus, Director-General of the World Health Organization, said: “WHO has today issued a medical product alert for four contaminated medicines identified in The Gambia that have been potentially linked with acute kidney injuries and 66 deaths among children.”
He added that the loss of these young lives is beyond heartbreaking for their families. The four medicines are cough and cold syrups produced by Maiden Pharmaceuticals Limited, in India.
As per reports, Ghebreyesus said at a press conference on Wednesday: “While the contaminated products have so far only been detected in the Gambia, they may have been distributed to other countries.”
He added that the global health body will ask all countries to detect and remove these products from circulation to prevent further harm to patients.